by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Mar 11, 2016 | Cancer Drugs, Drug Prices
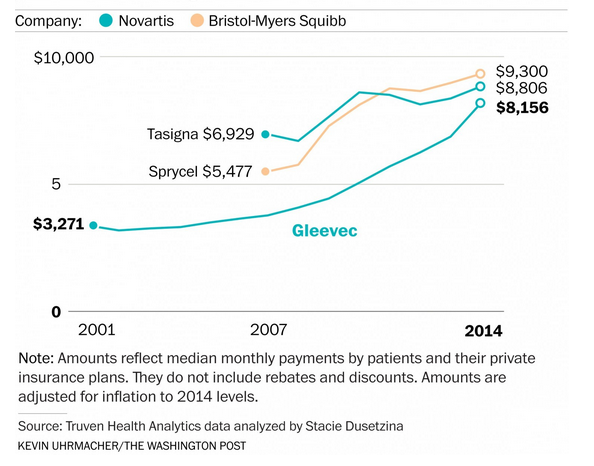
An article in the Washington Post yesterday covered the steep rise in the cost of a cancer medication called Gleevec, drug company Novartis’ brand name for imatinib, which is incredibly successful in treating people with chronic myeloid leukemia (CML). Despite its concern with public perception over price at the time, Novartis charged $26,400/year when the drug hit the market in 2001. With today’s price for Gleevec now $126,000/year it seems any concerns by Novartis have gone by the wayside (or did they really exist at all?). One would think and hope that new, similar products would bring down – or at least slow increases of – the price of Gleevec. After all, competition is supposed to drive down prices.
Not in the pharmaceutical industry, at least not until a drug goes off patent. The launch of therapeutically equivalent brand medications to treat CML actually coincided with Gleevec’s steepest price increases. Bristol-Myers Squibb launched Sprycel (dasatinib) in 2006 and Novartis launched a second generation drug called Tasinga (nilotinib) in 2007, both at much higher prices than Gleevec. In 2007, the monthly prices for Gleevec, Sprycel and Tasinga were $3,757, $5,477 and $6,929, respectively. The details can be found in the Post’s article but essentially Gleevec started to play “catch up” to its competitors.
In 2007, the price of Gleevec was 46% less than Tasinga. By 2014, the discount had shrunk to 12% with Gleevec’s price at $8,156/month compared to Tasinga’s price of $9,300/month. Let’s keep in mind that the newer drugs were shown to treat people that Gleevec could not treat, which would soften the argument that these are truly competing products. But soon after their introduction, Sprycel and Tasinga were found to successfully treat people with newly diagnosed cases of CML, to more directly compete with Gleevec, yet the latter’s price soared!
Source: Graph below from the Washington Post.
One source cited in the post’s article summed it up perfectly. A hematologist from the University of Chicago, Richard Larson, stated: “Ordinarily, you might think with three equally effective drugs on the market, the price should go down through competition, but it’s been a failure of the competitive pricing process.”
While the Washington Post’s article delved into the problem of out of pocket costs for Gleevec, it didn’t hammer home that the suffering in America is extreme when it comes to Gleevec’s drug price (and other medication prices as well). Gleevec is not a new topic on these blog pages, which has solicited comments from people and families crushed by drug costs. Yes, the irony is glaring: cancer medications that alleviate suffering also create suffering, too, in the form of cancer patients facing bankruptcy, feeling guilt, and causing anxiety.
It’s not just market failure – it is greed, too. I’ll let one American, Penny Kincaid, a commenter on this blog, bring it home:
“I guess I am luckier than many on Gleevec. I am paying 5% of the cost each month but the cost keeps going up. Our lives depend on this drug but still the cost is obscene. They praise themselves for creating [these] drugs for patients with [CML] but still we are forced to pay and many go broke. It is the cancer patient who has to pay up or die and they keep raising the cost. This is just wrong in so many ways and here we have the creators wanting to deny us the generic because they want that big money to keep rolling in. They should be ashamed of themselves.”
Tagged with: Bristol Myers Squibb, chronic myeloid leukemia, Drug Prices, Gleevec, leukemia, novartis, Sprycel, Tasinga
by Gabriel Levitt, Vice President, PharmacyChecker.com and Sam Werbalowsky, Pharmacychecker.com | Apr 5, 2013 | Drug Importation, Drug Prices, Politics
Americans interested in generic drug prices and pharmaceutical patent law have been closely following the U.S. Supreme Court as it hears arguments in a case over “pay-for-delay” – the practice of brand-name drug-makers seeking greater profits by paying off generic manufacturers to delay introduction of low-cost generic drugs. If “pay-for-delay” tactics are declared unconstitutional, then generics would reach pharmacy shelves faster, translating to lower prices for consumers, health insurers, and taxpayers.
The Court’s decision isn’t expected until June, but last Monday the Supreme Court of another country rendered another decision related to generics that may affect American prices. India’s Supreme Court ruled against the drug company Novartis’ patent claim on Gleevec, a cancer medication. Since India’s decision allows drug companies to continue manufacturing generic versions of Gleevec, called imatinib mesylate, prices will remain exceedingly low in India and low-income countries that import Indian pharmaceuticals.
So how much cheaper is generic Gleevec in Indian pharmacies than brand name Gleevec in American pharmacies? The New York Times reported that a one-year supply of brand name Gleevec in the U.S. is a staggering $70,000. The generic in India is only $2,500!
Additionally, even though Gleevec is under patent in other high-income countries like it is in the U.S., it is far less expensive internationally. At a local New York City pharmacy the price for 30 pills of Gleevec (400mg) is $6,980. The same brand name Gleevec (400 mg) from a Canadian pharmacy is just under $3,700. The same drug (but marketed by Novartis as Glivic), can be ordered online from Turkey for $2,979. That’s a potential savings of $4,000 a month! If you choose to buy Gleevec or any medication online, to protect your health, stick to verified online pharmacies, such as those approved by PharmacyChecker.com.
In the wake of the Indian Supreme Court’s decision, it would not be surprising if Novartis reacts by raising Gleevec prices here in America to bolster profits. Governments of other high-income countries probably won’t allow Novartis to raise prices on Gleevec, due to price controls. This is patently unfair to Americans, who should not have to pay so much more for the same medication than citizens of other high-income countries.
Tagged with: Gleevec, pay for delay, Supreme Court
by Gabriel Levitt, President, PharmacyChecker.com and Prescription Justice | Dec 5, 2019 | Drug Importation
A new Chinese law (in effect as of December 1, 2019) makes third-party
online platform medicine sales legal, appropriately ends a draconian definition
of counterfeit drugs, and effectively decriminalizes personal drug importation,
but with a lot of gray! The changes were part of a major overhaul of the Drug Administrative Law
of the People’s Republic of China (DAL). The previous linked to page is in
Chinese but you can use Google translate to read it in English or another
language. A summary in English can be found here:
Summary
of Revised Chinese Drug Administration Law
For supporters of online access to safe and affordable
imported medicines, this is kind of cool.
(more…)Tagged with: China, Gleevec, lu yong